Several vaccines with good efficacy exist for respiratory syncytial virus (RSV). Just launched and FDA-approved in 2023, the development of RSV vaccines is a significant advance in healthcare. RSV is very commonplace, and virtually all kids below the age of 2 have already had it at least once. RSV is highly contagious and circulates in the U.S. during the colder months.
Symptoms generally include a runny nose, breathing difficulty, loss of appetite, a cough that may turn into wheezing, and possibly fever. The infection in the small airways of the lungs can lead to bronchiolitis or pneumonia. Young children, seniors, people with weakened immune systems, and those with lung or heart disease are the most at risk for getting serious illness from RSV.

The impact of RSV in the U.S.
The impact of RSV is staggering. Every year, getting sick from RSV prompts 2.1 million outpatient visits among children under the age of 5. About 58,000-80,000 children are hospitalized, and 100-300 children die from RSV-related complications.
The impact on the elderly is even greater, even though until recently RSV was mostly considered a children’s disease. Every year 60,000-160,000 adults over the age of 65 are hospitalized, and 6,000-10,000 die due to RSV-related complications.
Since RSV is so common and potentially deadly, it is surprising that a vaccine did not occur for RSV until earlier this year. After over 60 years of efforts, a string of failures, and many challenges, RSV vaccines were finally released, approved by the FDA, and recommended by the CDC in 2023.
Why it took so long to get RSV vaccines
According to the WHO, a vaccine against RSV was first developed in the 1960s and was given to infants. But the vaccine caused severe (and in two instances, lethal) inflammatory response in the lungs when the infants first contracted RSV after their immunization. This failure hindered and discouraged the development of new RSV vaccines for decades.
In the 1980s, Paul Glezen, an immunologist and microbiologist at Baylor College of Medicine tested blood samples from RSV-afflicted infants and was able to correlate good recovery from RSV infection with better anti-RSV antibodies in an infant’s blood. This proved that antibodies in an infant’s blood, either made on their own (after a previous RSV encounter) or obtained from the mother’s body during pregnancy, can protect the infant from the worse effects of RSV. This finding proved the value of pursuing research in RSV vaccines, which can provide infants with antibodies to protect them from severe lower respiratory tract disease when their lungs are still developing.
Over the next 40 years, as our understanding of how viruses work improved, scientists found better targets on the virus that antibodies could recognize and tag, marking the virus for destruction through the cellular machinery. The current vaccines use this new understanding to develop safe and effective RSV vaccines, which have great effects on preventing serious illness and hospitalization from RSV. The effects from one dose of vaccine lasted for one or two RSV seasons.
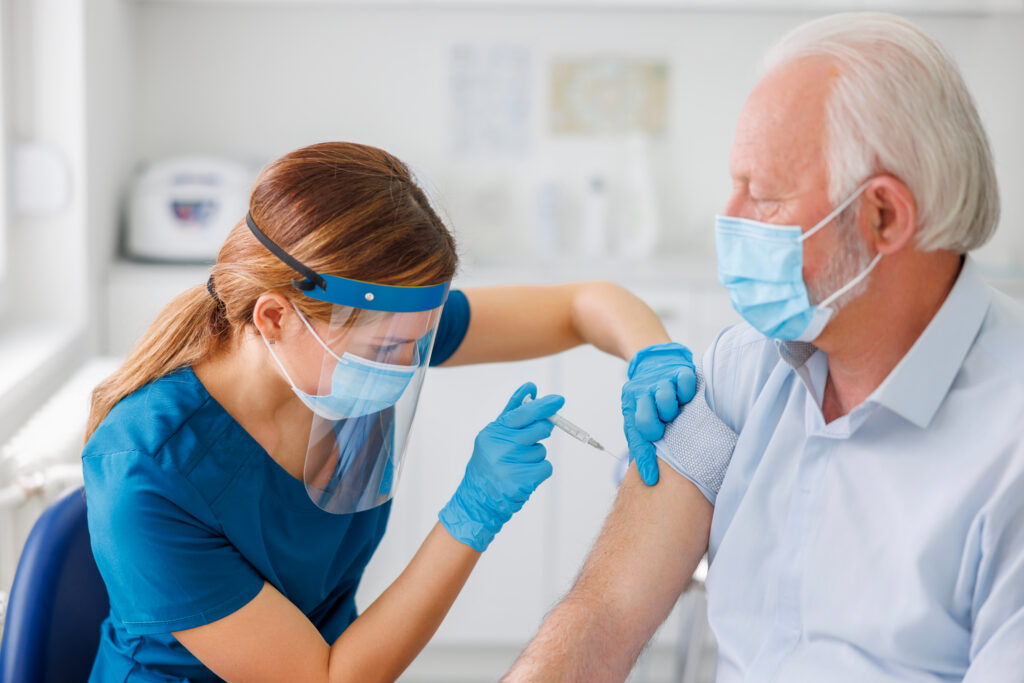
Currently available RSV vaccines
RSV vaccines for vulnerable seniors
The CDC recommends that adults 60 years of age or older have the option to receive a single dose of RSV vaccine. The FDA-approved vaccines for this age group are GSK’s Arexvy and Pfizer’s Abrysvo.
RSV vaccines for infants younger than 6 months old
For this age group, the CDC recommends two possible routes of RSV immunization.
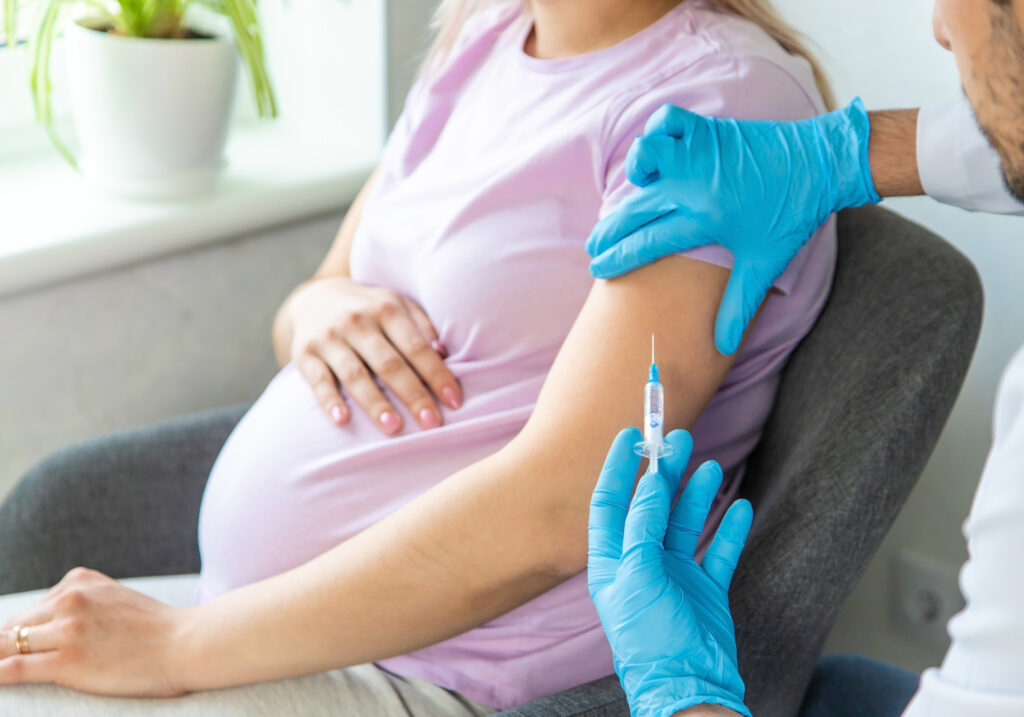
- RSV vaccine that is given to pregnant people through weeks 32-36 of pregnancy. If the baby is born 2 weeks after the mother receives the vaccine, they are expected to have sufficient antibodies in their blood for some months to protect them from the worst effects of RSV infection. The only vaccine approved for this category is Pfizer’s Abrysvo.
- RSV vaccine that is given to infants if their mothers did not receive RSV vaccination, or who were deemed vulnerable by their pediatricians based on factors such as premature birth, chronic lung or heart disease, or a weakened immune system. The current vaccines recommended by CDC in this category are Nirsevimab (Beyfortus) for infants 6 months old or younger, and Palivizumab (Synagis) for vulnerable children 8-19 months of age.
How effective are the current RSV vaccines
Arexvy. In adults 60 years of age or older with healthy immune systems, Arexvy (single dose) was 83% effective in preventing RSV-related lung infection in the first RSV season, and 56% effective in the second RSV season.
Abrysvo in seniors. In adults 60 years of age or older with healthy immune systems, Abrysvo (single dose) was 89% effective in preventing RSV-related lung infection in the first RSV season. The results from the second season have not been released yet.
Abrysvo in pregnant people. In the clinical trials, pregnant individuals who got the vaccine provided their infants immunity from RSV after birth. When approximately 3500 pregnant individuals received one dose of Abrysvo, the vaccine reduced the risk of severe lower respiratory tract disease (LRTD) by 81.8% within 90 days after birth, and 69.4% within 180 days of birth.
In a subgroup of pregnant individuals, who were immunized with Abrysvo during 32-36 weeks of pregnancy, it reduced the infant’s risk of LRTD by 34.7% and severe LRTD by 91.1% within 90 days of birth. Within the first 180 days of birth, the vaccine reduced the risk of LRTD by 57.3% and severe LRTD by 76.5%.
Nirsevimab. In clinical trials, Nirsevimab was 79% effective against LRTD. The vaccine was 80% effective at preventing hospitalization in infants entering their first RSV season.
Palivizumab. In clinical trials, Palivizumab prevented serious LRTD caused by RSV. The vaccine had 45-82% efficacy in preventing RSV-related hospitalizations in high-risk infants.
RSV vaccines are not for everyone
RSV vaccines may not be suitable for every individual. Always talk to your child’s pediatrician, your ob-gyn, or your physician before getting the RSV vaccines. A doctor will take into account your health history and discuss any side effects of the vaccines that may apply to you to see if you can benefit from RSV vaccination.
Current challenges to the adoption of the RSV vaccines
In the wake of COVID-19, public health officials worry that the public may be feeling “vaccination fatigue”. After all, the RSV vaccine is one more shot to get in addition to the annual flu shot, and the updated COVID-19 vaccine booster. Luckily, the CDC says that in most cases, all three vaccines (RSV vaccine, flu vaccine, and the COVID-19 booster) can be taken at the same time. The ability to get all three vaccines at the same appointment may increase the RSV vaccine’s adoptability.
What comes next in the world of RSV vaccines
The vaccines discussed here are only the first ones out of the gate to combat RSV. As of 2021, there were at least 30 other RSV vaccines in various stages of clinical trials. Some of these are meant for pregnant women to boost their immunity against RSV and provide protection for their babies. Others are targeted towards one of the other two most vulnerable groups- children, or the elderly.
Not all vaccines use the same approach to target the virus. Depending on the success of these clinical trials, additional RSV vaccines may become available in the coming years. With more data about a vaccine’s mode of action, effectiveness, and side effects, physicians and pediatricians in the future may be able to offer a tailored recommendation best suited for individual patients.
The information provided in our blog posts is for informational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this blog.
 What are the symptoms of bird flu?
Prev post
What are the symptoms of bird flu?
Prev post





